Water electrolysis
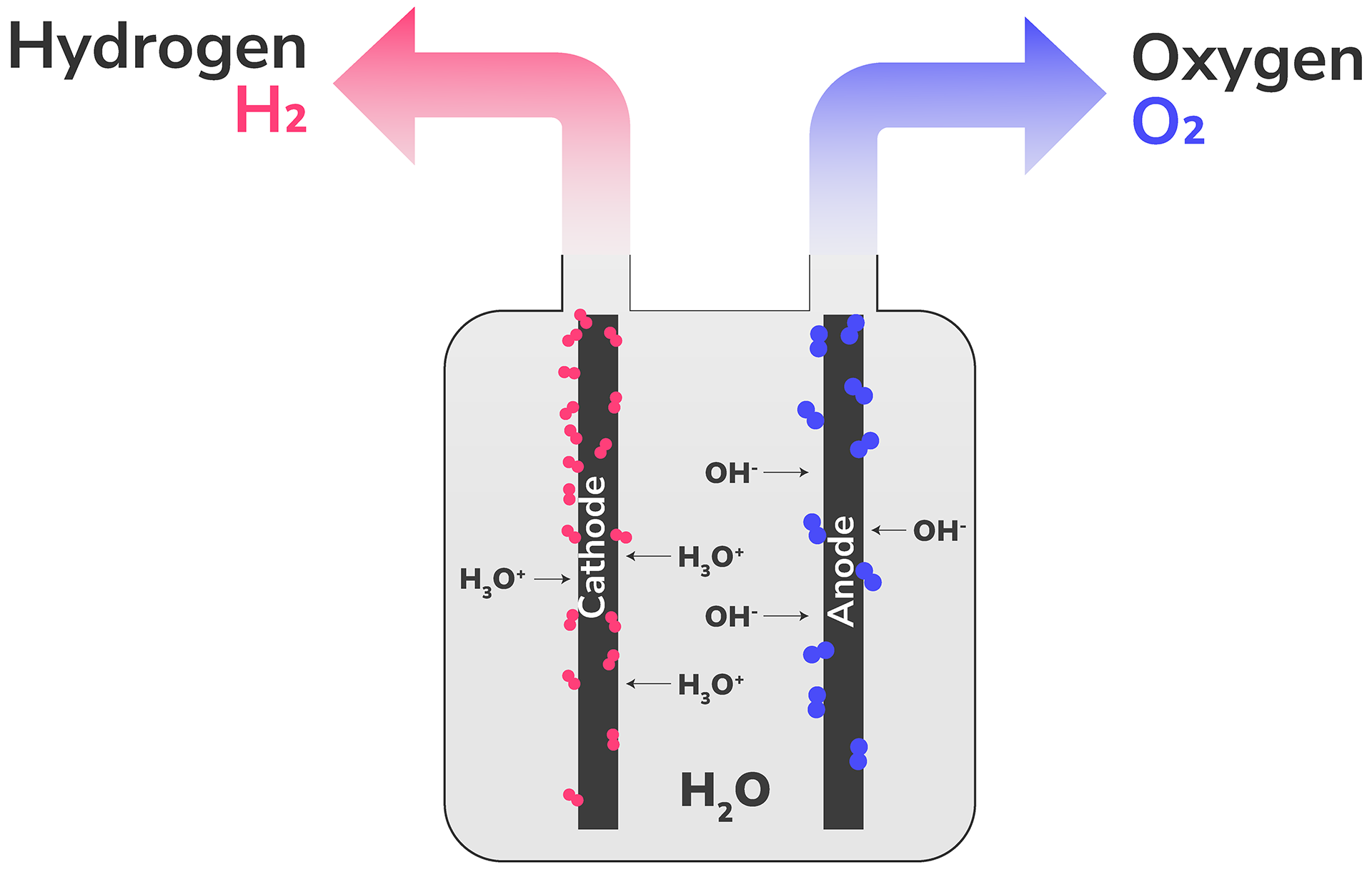
A method for decomposing water molecules (H₂O) into hydrogen (H₂) and oxygen (O₂) by applying electrical energy.
Source: IRENA. Innovation trends in electrolysers. 2022
Main electrolyzers and their characteristics
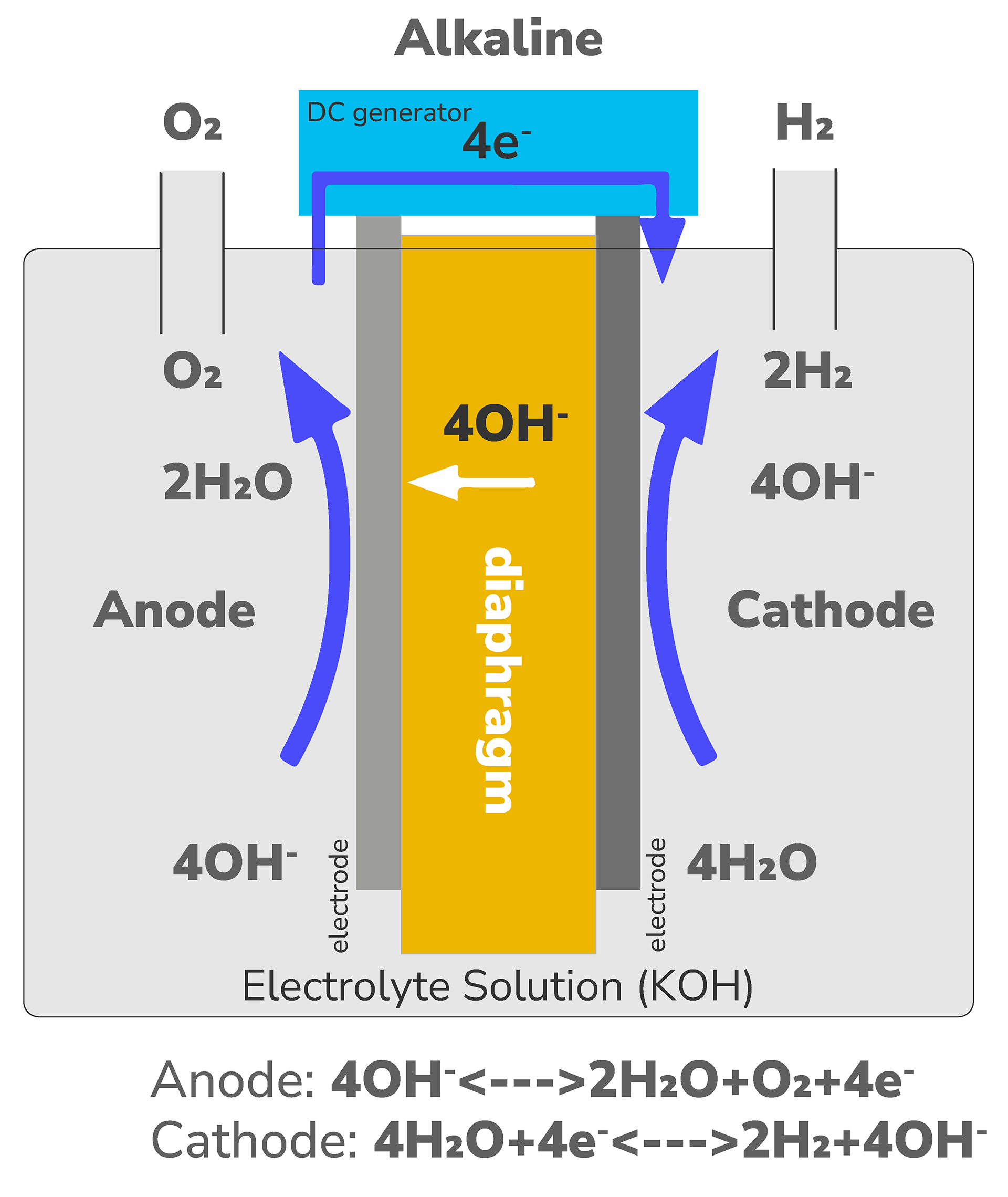
Alkaline Electrolyzer
- Technology implemented on an industrial scale
- Extensive operational experience (having been in use for nearly a century, providing substantial operational knowledge)
- Low specific investment costs (110-1800 USD/kW)
- Limited operational flexibility, requiring a constant load
- Compact design due to high current density
- Compact design due to high current density

Proton Exchange Membrane (PEM) Electrolyzer
- Capable of operating with variable loads
- Able to remain inactive for extended periods without necessitating special procedures for reactivation
- Requires fewer personnel
- Entails higher specific investment costs (2800-5600 USD/kW)
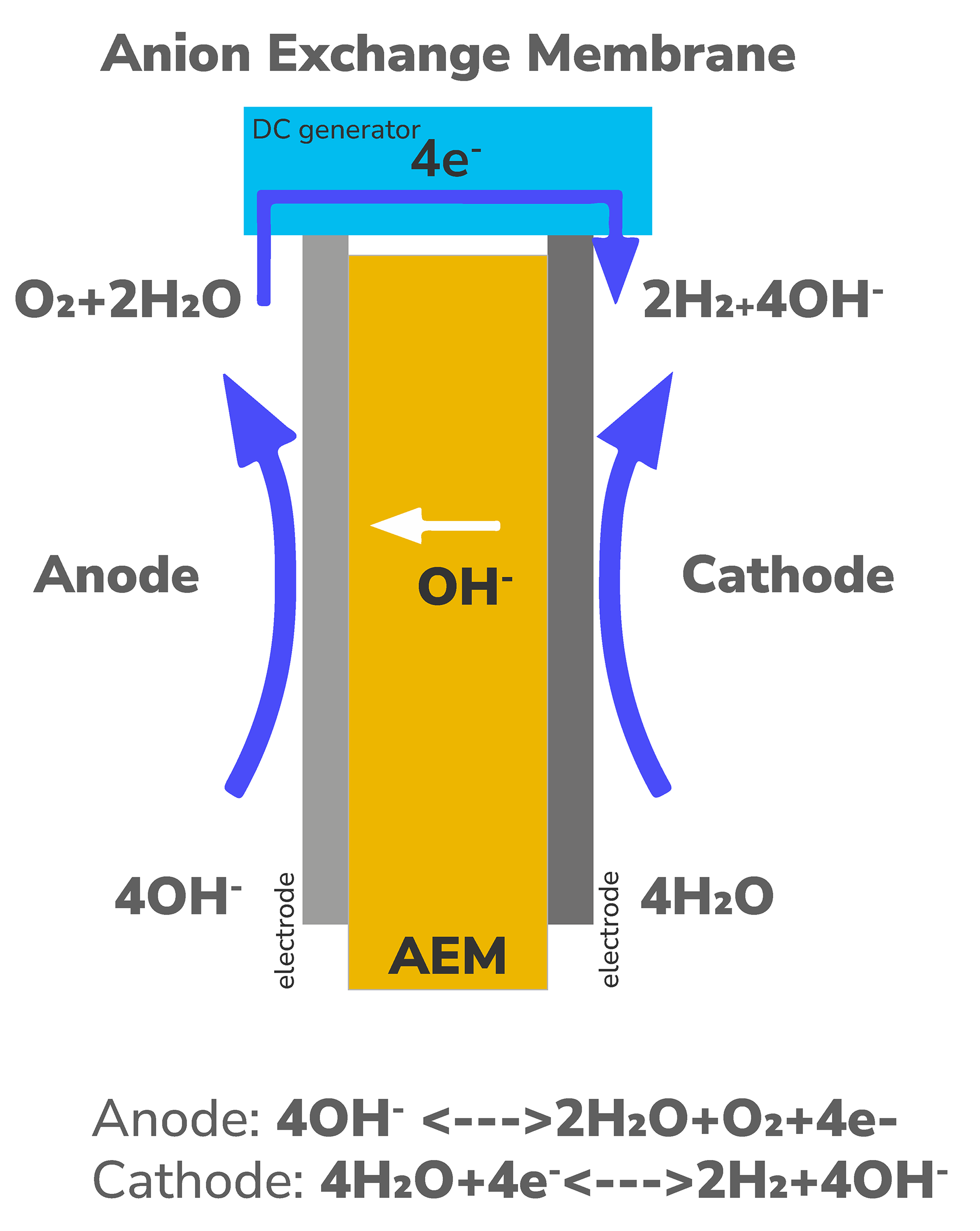
Anion Exchange Membrane (AEM) Electrolyzer
- Potentially competitive with alkaline electrolyzers in terms of cost
- Flexible in operation, capable of ramping up and down with renewable energy sources
- Concerns regarding degradation rates

Solid Oxide Electrolytic Cell (SOEC)
- High temperature electrolyzer with the highest efficiency potential, well-suited for challenging industrial processes requiring decarbonization and utilizing waste heat
- High temperature electrolyzer with the highest efficiency potential, well-suited for challenging industrial processes requiring decarbonization and utilizing waste heat
- Lower durability
Source:
Adaptado de:
IRENA, Green Hydrogen Cost Reduction: Scaling up Electrolysers to Meet the 1.5⁰C Climate Goal. 2020
IRENA. Innovation trends in electrolysers. 2022
Main characteristics of electrolyzers
| 2020 | 2050 | |||||
|---|---|---|---|---|---|---|
| Alkaline | Proton Exchange Membrane | Alkaline | Proton Exchange Membrane | Anion Exchange Membrane | Solid Oxide Electrolytic Cell | |
| Cell Pressure (bar) | <30 | <70 | >70 | >70 | >70 | >20 |
| Efficiency (System) (kWh/kg H2) | 50-78 | 50-83 | <45 | <45 | <45 | <40 |
| Efficiency (System) (kWh/kg H₂) | 60 | 50-80 | 100 | 100-120 | 100 | 80 |
| CAPEX Stack >1 MW [USD/kWel] | 270 | 400 | <100 | <100 | <100 | <100 |
| CAPEX Stack >1 MW [USD/kWel] | 500-1400 | 1100-1800 | <200 | <200 | <200 | <300 |
Source: Adaptado de IRENA. Green Hydrogen Cost Reduction: Scaling up Electrolysers to Meet the 1.5⁰C Climate Goal, International Renewable Energy Agency, Abu Dhabi. 2020
Water availability
Single (left) and double (right) drains of the Colombian hydrographic network.
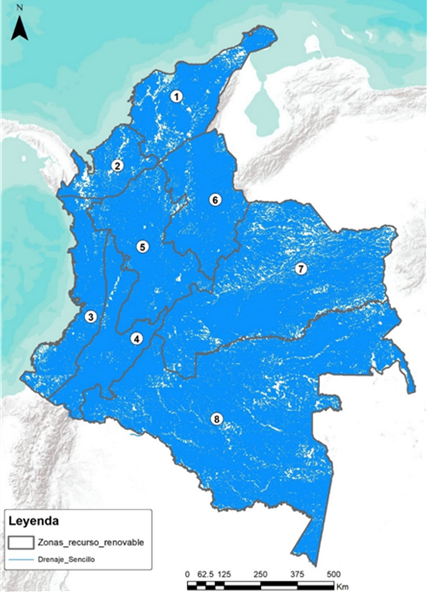

Source: GIZ Colombia. Estudio técnico, económico e identificación de hubs de hidrógeno verde en Colombia. 2023.
Simple drainage is defined as a watercourse of natural origin with either permanent or intermittent flow. This category encompasses all rivers, streams, creeks, and other watercourses that do not receive contributions from other surface water flows. Double drainages are defined as natural water currents that flow continuously, possess a determined flow, and discharge into the sea, a lake, or a river; in the latter case, they are referred to as tributaries. In general, they constitute the main hydrographic network.
In general, there are no areas in the country located more than 100 km from a double water source, i.e., one with the capacity to supply a hydrogen plant. However, it should be noted that these water resources are highly susceptible to climate change and must be managed efficiently to ensure their sustainability in the medium and long term, thereby avoiding significant negative impacts on communities.